 Real-Time Magazine analysis reveals a concerning issue almost no one talks about: serious side effects and deaths of breastfed infants whose mothers received the COVID-19 vaccine.
Real-Time Magazine analysis reveals a concerning issue almost no one talks about: serious side effects and deaths of breastfed infants whose mothers received the COVID-19 vaccine.
Participated in the analysis of the reports: Shani Cohen, attorney Galit Potachnik and Shirley van der Veldt
Despite the lack of clinical studies, and even animal safety studies, health authorities did not hesitate to give the green light to the administration of Pfizer’s and Moderna’s mRNA vaccines to pregnant and lactating women a few weeks after they themselves stated that they did not have such studies. In Israel, the MOH even made a U-turn in a record time of four days, when already on December 20 a recommendation was issued for pregnant and lactating women to get vaccinated. Nevertheless, the issue of vaccine safety for breastfed infants is hardly discussed.
An analysis conducted by the Real-Time Magazine team reveals that at least 648 cases of side effects in infants vaccinated with the COVID-19 vaccines – most of them with the mRNA vaccines, including deaths and life-threatening side effects, have been reported to the US VAERS system.
The most common serious events were life-threatening bleeding; anticholinergic syndrome; Liver problems; anaphylactic shock; neuroleptic syndrome; neurological side-effects such as convulsions or encephalitis; and Hypoglycemia. In most of the reported cases, several life-threatening side effects were recorded in the same baby.
“I was breastfeeding my son at the time. I pumped within an hour of receiving the shot. My 5 month old son nursed the night following the shot and later ate the pumped milk that I acquired the same day of the shot. 11 days later he was found unresponsive during his nap at daycare. It is not clear how long he was unresponsive for until found by babysitter. He was immediately rushed to the hospital and doctors were able to get his heart beating again with excessive effort. Organ damage was extensive and he had no brain function, he did not recover. He passed away 13 days after I received the shot”. The VAERS report says: “Died? No Recovered? No”.
This shocking description, a report by a mother who lost her baby, a five-month-old breastfed infant, 13 days after receiving the Pfizer COVID-19 vaccine and breastfeeding him, is taken from the VAERS system – the American Adverse Event Monitoring System (case number 1505306).
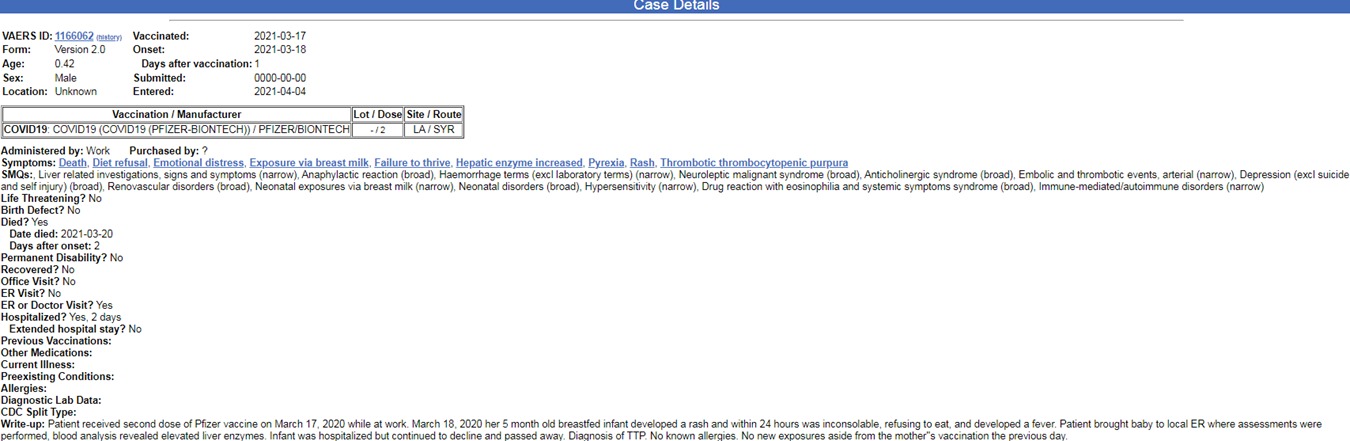
As can be learned from the report, the mother received the vaccine on January 28, very shortly after the FDA recommended that pregnant and lactating women receive the COVID-19 vaccine. Alarmingly, this is not the only report of a baby being harmed or dying after being exposed to the mRNA vaccine through breastfeeding. In another chilling report (case number 1166062) by a mother, dated March 17, 2021, a case of another breastfed baby, also five months old, who died just one day after his mother was vaccinated during a work day is described. The next day, March 18, 2021, the baby felt ill, developed a fever and a rash, refused to eat and cried non-stop. The write-up said:
“Patient brought baby to local ER where assessments were performed, blood analysis revealed elevated liver enzymes. Infant was hospitalized but continued to decline and passed away. Diagnosis of TTP. No known allergies. No new exposures aside from the mother’s vaccination the previous day”.

What was the cause of the two babies’ deaths? Is it possible that their deaths were related to the fact that their mothers were vaccinated a few days earlier, and that they were exposed to the mRNA vaccine through breastfeeding? Amazingly, although health authorities around the world began vaccinating breastfeeding women shortly after the FDA granted Pfizer the emergency permit, without having carried out studies in this sensitive population or even toxicity studies in animals, and even though more than a year and a half have passed since then and many millions of breastfeeding women around the world have already been vaccinated – the health implications of the mRNA vaccine on nursing babies are almost never considered or discussed significantly.
In order to try to get an answer to this question, even if only a partial one, Real Time Magazine conducted an analysis of the reports to the VAERS system on adverse events among nursing infants whose mothers were vaccinated. From the analysis, which is a continuation of the previous one we conducted on VAERS reports on babies who were themselves vaccinated, it appears that the stories presented above represent a phenomenon with a very large scope. As mentioned, at the time the analysis was conducted, there were 648 reports registered in the VAERS system on babies who suffered adverse events after being exposed to COVID-19 vaccines through breastfeeding. In some cases, these are serious and even life-threatening adverse events, of which, 3 confirmed deaths have been reported, and in other cases, it is not clear what happened to the babies – did they recover, do they continue to suffer from health problems, or whether they died.
Side effects in a variety of body systems
For the purpose of the analysis, we initially performed a search through the system itself, using keywords such as Breastfeeding, Nursing, etc. In this search, 85 reports were displayed in the VAERS system. Surprisingly, an attempt to reach a result through a Google search led to a VAERS page displaying 629 reports of adverse events following exposure to COVID-19 vaccines through breastfeeding. During the process, the number of reported cases increased, and as mentioned, at the end of our data collection, on August 5, there were 648 reports. In view of the centrality of the mRNA vaccines, and the fact that this is a new technology, the analysis focused on these vaccines only.
After omitting reports of other types of COVID-19 vaccines, such as Jensen and AstraZeneca, 606 reports were identified, of which 435 were reports of side effects following the Pfizer vaccine and 171 reports following the Moderna vaccine. For the purpose of the analysis, the reports were placed and presented in an Excel table.
Our magazine team read each of the reports and analyzed them according to the types of side-effects and their seriousness, the time of their appearance after the vaccine, the duration of the side-effects, the ages and gender of the babies, and the manufacturer of the vaccine.
We analyzed both the data and information presented in the closed categories in each of the reports, and in the free text in which the case was described. Regarding reports that were not clear, the team members cross-examined them, to try to clarify the case. It should be noted that in some cases there was ambiguity and confusion in the case descriptions, in a large part of which the information is missing, such as the gender of the baby, its age, or the batch number – problems that can probably be attributed to the fact that the VAERS system is a self-reporting system. For example, in some cases it was not possible to clearly understand whether the mother was the one who experienced the side effect or her baby. It should also be noted that in several cases, when we returned to certain reports, we found that the report had changed – usually, descriptions and side effects were added to the initial report. In several reports, parents expressed fear regarding the baby’s side effects, and raised questions about the vaccine and other potential side effects.
The findings of the analysis indicate that the reports represent a variety of side effects in various body systems, with the age range of the nursing infants affected following their mothers’ vaccination being from the moment of birth to approximately 3 years of age. Some of the side effects were prolonged and life-threatening. Some babies experienced more than one side effect. In many cases it was reported that the baby did not recover and that the case could not be followed. In other cases it is noted that the patient was not included in the experiment.
Serious Side-Effects
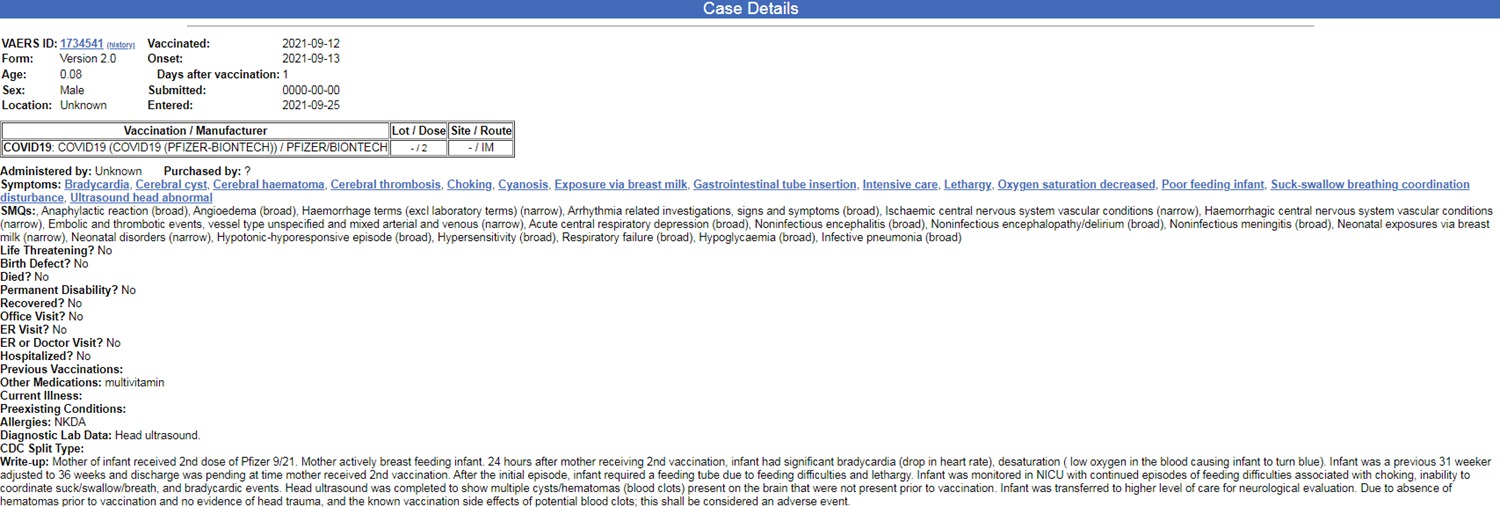
The analysis identified three definite reports of deaths (reports number 1505306; 1166062 1712010). As noted, in other cases it is not clear from the report whether the baby survived. For example, in one case (case number 1734541) a 28-day-old baby was exposed through breastfeeding to the second Pfizer vaccine dose that his mother received. 24 hours after the vaccine was given, the baby was in a state of anaphylactic shock, bradycardia (low heart rate), low blood oxygen saturation that made his skin appear blue, and respiratory collapse. The baby also suffered bleeding in the brain, with a brain scan showing blood clots. Since he was unable to nurse, they had to feed him through tubes. Although in the closed categories it was stated that he did not die – in the report itself it was stated that he did not recover.
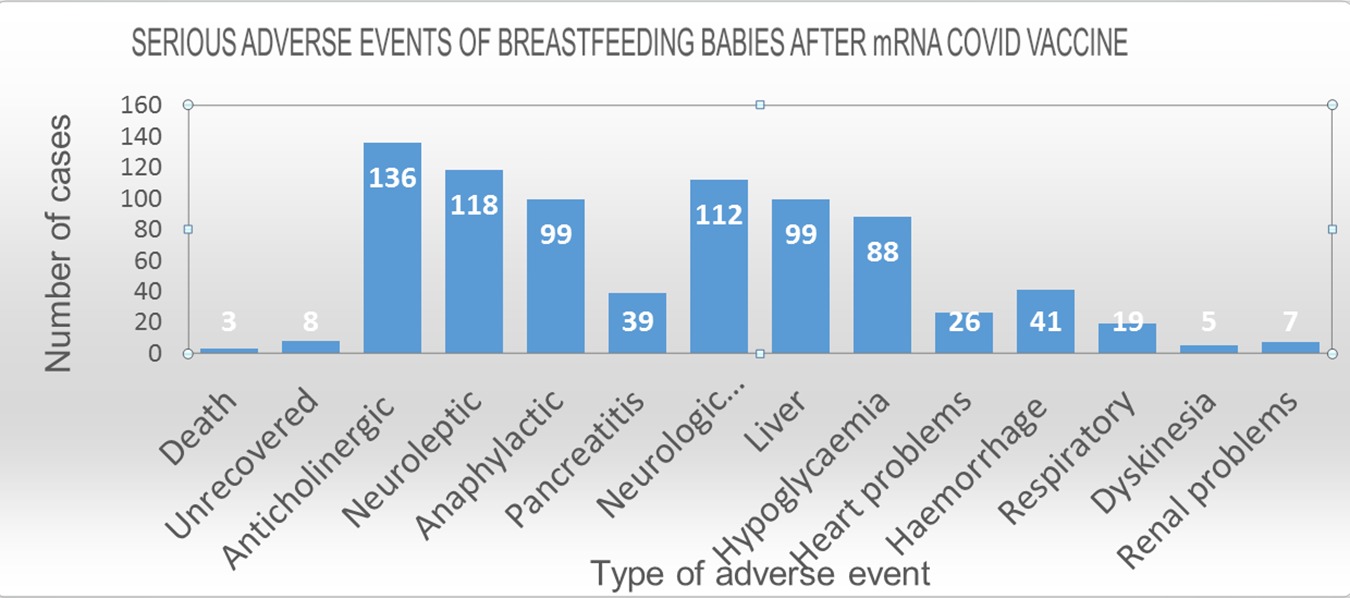
Some of the side effects identified in the reports were particularly common. As can be seen from the chart below, the most common serious side-effects were anticholinergic syndrome (136 reports); neuroleptic malignant syndrome (118 reports); neurological phenomena (112 reports); Liver damage (100 cases), anaphylactic shock (100 reports) and hypoglycemia (88 reports). This finding is similar, and in fact almost identical to the common side-effects identified in our previous analysis published in Real-Time Magazine, of reports in 58 infants who were themselves vaccinated – a similarity that reinforces the likelihood of causal relations between these side-effects and the vaccine.
Additional adverse events identified in the present analysis of the nursing babies’ reports include pancreatitis (39 reports); heart problems (26 reports); respiratory problems (19 reports); renal problems (7); and dyskinesia (movement disorders) (5).
Anticholinergic syndrome – the most common serious side-effect, which was identified in 136 of the reports, is anticholinergic syndrome – a syndrome resulting from a blockage of the binding sites of the neurotransmitter acetylcholine, which can lead, among other things, to coordination problems, an increase in body temperature, and an increase in heart rate to the point of collapse. This syndrome is well known among adults who take various medications, such as antipsychotics, antihistamines, etc., and is not considered life-threatening for them. However, in children and toddlers, the syndrome may rapidly deteriorate and require intensive care. In a study that examined the neurological effects of COVID-19 vaccines, published in January 2022 in Cureus, it was found that the Pfizer vaccine has the greatest risk of leading to this syndrome, with the risk increasing the lower the age of the vaccinated. The researchers hypothesized that the syndrome may be related to the fatty envelope in which the mRNA is found – which distinguishes Pfizer’s vaccine from other COVID-19 vaccines, and perhaps therefore, the use of this vaccine increases the risk.
Neuroleptic malignant syndrome (NMS) – This syndrome, the second most common serious side-effect, was identified in 118 reports. It is a life-threatening syndrome, which may manifest itself in an accelerated heart rate, an increase in liver enzymes, hypertension, epileptic convulsions, and more, and may involve the destruction of nerve cells in the brain to the point of brain death. It is usually caused after taking antipsychotic drugs. This side-effect was almost never documented in the literature in connection with the COVID-19 vaccine, but in November 2021, an article in the American Journal of Emergency Medicine documented a case description of a 74-year-old woman who developed the syndrome after taking the COVID-19 vaccine, and was hospitalized in the intensive care unit. The researchers warned that the activation of the immune system associated with these vaccines can have a huge impact on the elderly, and that awareness of neuroleptic malignant syndrome should be raised as a potential complication of the COVID-19 vaccines, and emergency room doctors should be provided “with the patient’s vaccination history to properly diagnose this problem, and possible side effects others of this new one”. In light of this case description, which is defined in the article as apparently rare, the finding from our analysis of reports of nursing infants in the VAERS system indicates one of two things: either the risk of this side-effect is particularly high in small babies, compared to the adult population, or the real risk of its prevalence is simply not documented.
Liver injury and anaphylactic shock – Regarding each of these side-effects, which appear to be the third most common ones in our analysis, 99 reports were identified. In light of the many cases of mysterious liver problems among children around the world published by health authorities and in the media, any case of liver damage must be examined seriously, especially when it comes to a relatively high incidence among nursing babies. From an animal study conducted by Pfizer itself, it appears that the lipid nanoparticles (LNPs), the ones that contain the mRNA code reserved for the cells to produce the spike protein, do not remain at the injection site, but penetrate the bloodstream and through it reach a variety of tissues and sites in the brain, including the liver. The research, called “Biodistribution Research”, was defined by the company as confidential (“Pfizer confidential”), but was revealed a little over a year ago thanks to access granted by the Japanese Medicines Agency to a number of researchers. It should be noted that the study has been deleted from the web, but can be found in the Internet Archive.
An anaphylactic shock is a life-threatening allergic condition that requires immediate treatment. It can damage the respiratory tract and cause dizziness, fainting, and even death.
Neurological side-effects such as convulsions, or inflammation of the brain (encephalitis) – 112 cases were identified. These side effects can cause serious damage to the brain.
Hypoglycemia – 88 cases of hypoglycemia were identified in the VAERS reports – a very low blood glucose level, a condition that in babies can quickly deteriorate into an irreversible condition and even death. Glucose is used in the process of cellular respiration. When brain cells are not supplied with glucose, this vital process is impaired and irreversible brain damage occurs. The relationship between the mRNA vaccines and hypoglycemia among diabetic patients treated with anti-diabetic drugs and/or insulin was documented by researchers in Western Saudi Arabia, and in a study registered in October 2021 on the American Federal Clinical Research website (the study has not yet been published), they are trying to examine the nature of this relationship. The researchers hypothesized that the preservative polyethylene glycol (PEG) contained in the mRNA vaccines may be the cause of the hypoglycemic effects.
Bleeding – 41 of the reports describe cases of bleeding, some in the stool or urine, some in vomiting and even bleeding from the eyes.
39 reports describe cases of pancreatitis – acute inflammation of the pancreas, which can cause fever, rapid pulse, drop in blood pressure, and even respiratory failure and/or kidney failure. In addition to these serious effects, 26 cases of heart problems of varying seriousness have been reported among nursing babies, including heart rhythm disturbances, heart muscle enlargement, and a hole in the heart, as well as two cases of myocarditis and pericarditis.
Side-effects with mild and moderate degrees of severity
In addition to these serious side effects, the analysis of the reports also identified less serious symptoms, including vomiting, fatigue and sleeping disorders, diarrhea, nursing problems, refusal to eat, and even several cases of green milk or green feces. Although these side effects are seen as relatively minor problems that appear even after routine vaccinations, it is important to address them, as they can be symptoms of more serious problems. Also, 62 reports of rash were identified. In babies, this symptom can be considered mild and transient, yet in some cases, it can be a symptom of a life-threatening allergy.
Why were the lessons from the thalidomide and the DES affairs not learned?
As noted, shortly after the FDA granted Pfizer the emergency permit for its COVID-19 mRNA vaccine, the world’s health authorities reversed their previous decision not to recommend vaccinating pregnant and breastfeeding women, issued in early December. The original decision – not to recommend the vaccine to this vulnerable population, was based on the fact that pregnant and breastfeeding women were not included in the Pfizer and Moderna clinical studies, as can be learned, for example, from the Pfizer study protocol on the American federal clinical research website. In fact, to this day, the vaccine leaflet that appears on Pfizer’s website, states: “It is not known whether COMIRNATY is excreted in human milk. Data are not available to assess the effects of COMIRNATY on the breastfed infant or on milk production/excretion”.

Moreover, as several researchers have warned, the safety of the vaccine during breastfeeding has been lacking in evidence even in animal studies. For example, in the British government’s official guidelines document regarding the COVID-19 vaccine, it was explicitly stated that the studies on the toxicity of the vaccine in animals were not completed at the time the vaccine received the Emergency Use Authorization (EUA), and that it is not known whether the mRNA vaccine has an effect on fertility, pregnancy or breastfeeding.

When it comes to new drugs, pregnant and lactating women are defined as one of the most vulnerable and sensitive populations. This is due to the potential effects of the drugs on the fetus and the baby. The case that shook the world and illustrated how dangerous a hasty approval of a drug could be in this vulnerable population, without studying its effects on pregnant/lactating women in proper clinical studies, is the thalidomide scandal. In October 1957, the German pharmaceutical company Grunenthal began marketing the drug, which until then had been marketed as a sedative – as an over-the-counter medicine for pregnant women suffering from nausea. Patent usage rights were sold to manufacturers in various countries, including Canada, Great Britain, and also Israel. However, within a short time, more and more cases of babies being born with severe deformities, most of them in the limbs, including missing, short and deformed limbs, began to appear. Only in 1961, when it became clear beyond any doubt that birth defects were a side-effect of thalidomide, its distribution and marketing stopped. Following this affair, the European Medicines Administration changed its procedures, and it now requires teratogenicity testing of all medicines before marketing them. In the United States, thanks to Dr. Frances Kelsey, a physician who worked at the FDA, who insisted on not allowing the drug to be widely marketed, many injuries were avoided, and Dr. Kelsey was awarded the highest civilian honor by then-President John Kennedy.
Another case, which further emphasizes how critical it is to exercise extreme caution when it comes to vaccinating pregnant and lactating women, is the DES affair. DES (Diethylstilbestrol), a drug based on a synthetic estrogen manufactured by Eli Lilly, was given between 1938 and 1971 to pregnant women who had miscarriages or premature births, to prevent further miscarriages. At that time, it was believed that these problems could be caused by low levels of estrogen in the woman’s body. The DES was administered to millions of women in the United States during this period.
Unlike thalidomide, no visible medical problems were observed in the babies after birth. Only years later, when they grew up, it became clear that the girls whose mothers took the drug during pregnancy had an increased risk of suffering from several types of cancer, including adenocarcinoma of the endometrium and breast cancer; structural changes in the reproductive system, difficulties in conceiving, ectopic pregnancies, and other fertility and pregnancy problems. In 1971, the FDA advised doctors to stop prescribing the drug during pregnancy, DES was removed from the shelves and the issue was exposed to the public. The most important lesson from this affair was that the side effects of a drug that is taken during pregnancy may be discovered much later, only years later.
But it seems that all the lessons learned from the thalidomide and DES scandals were thrown away with the approval of the COVID-19 vaccines. As early as July 2021, the CDC announced that the Pfizer vaccine is safe for pregnant and lactating women. This, the CDC claimed, is based on “data from reporting systems” and based on “studies”. However, none of the studies cited in the list of sources in the document tested the safety of the vaccine during breastfeeding. Although the list of 23 sources includes several studies that dealt with the effects of the vaccine during breastfeeding, these studies concentrated on the issue of effectiveness (measured by antibody levels in breast milk), but not to the safety of the vaccine for the nursing infant. Nevertheless, in August 2021, the CDC issued a press release in which Dr. Rochelle Walensky, head of the CDC, was quoted as saying that the vaccine is safe for pregnant and lactating women, and that the CDC encourages all pregnant and lactating women to get vaccinated.
In fact, most of the studies on breastfeeding mothers conducted to date were relatively small and included a few dozen breastfeeding women, with most of them still focusing on the issue of effectiveness only, based on antibody levels in breast milk. For example, in a study published in January 2022 in Frontiers in Immunology, researchers from Singapore tested blood and milk samples from 35 nursing mothers. In addition to the antibodies, the researchers reported that they also detected low amounts of the whole mRNA in 71% of the mothers’ blood samples, and in 13% of the breast milk samples, and for some also in their breast milk. According to them, “The presence of intact vaccine mRNA in both sample types in our study highlights the stability and persistence of the vaccine mRNA nanoparticle within the bloodstream which may lead to infrequent transfer into breastmilk”. They also noted that the systemic spread of lipid nanoparticles from the mRNA envelope has already been previously demonstrated in animal studies.
In March 2021, a study conducted in collaboration with Tel Aviv University and Lis Hospital for Women and Maternity was uploaded to medRxiv. The study was conducted in January-February 2021, close to the launch of the COVID-19 vaccine rollout in Israel, and examined the antibody levels in the blood and breast milk at four-time points after the administration of the Pfizer vaccine. Yet, as it turns out, the study did not examine the safety of the vaccine or looked into side effects that appeared following vaccination in the infants or the mothers. Its sole purpose was to determine whether the COVID-19 vaccine is effective in creating antibodies in breast milk, and the qualities of these antibodies (that is, whether they are antibodies that can neutralize the virus). Moreover, the study included only ten lactating women.
In another study on 1,784 lactating women, published on Pre-Prints, researchers from Singapore and Malaysia examined the effects of mRNA vaccines and compared them to those of vaccines not based on this technology. The research findings showed that 6.2% of the mothers (110 mothers) reported that they stopped breastfeeding, with the vast majority receiving mRNA vaccines (97.2%). The reasons for stopping breastfeeding were instruction or recommendation from medical professionals, or problems with breastfeeding such as a sharp decrease or fluctuations in milk supply.
Pfizer itself announced in February 2021 a clinical trial in pregnant and lactating women. However, the results of this study have not been published to date.
Israel was the first to vaccinate pregnant and lactating women
Israel was the first country in the world to recommend vaccinating pregnant and lactating women, when the speed with which the Ministry of Health reversed the original recommendation, not to vaccinate this population, was surprising even on a global scale. On December 16, with the start of the vaccination rollout in Israel, the Ministry of Health issued an “Information Sheet for Vaccination Against the New Corona Virus”, with the instruction that children under the age of 16, as well as pregnant and lactating women, should not be vaccinated. The explanation: “This, since the vaccine has not yet been tested on children under the age of 16 or on pregnant and lactating women”.
Yet four days later, on December 20, 2020, it seems that science has advanced by leaps and bounds, as the Ministry of Health, in collaboration with the Israeli Association of Obstetrics and Gynecology, issued new guidelines, according to which it is now recommended to vaccinate pregnant and lactating women.
Moreover, less than a month later, on January 18, 2021, the Ministry of Health, in collaboration with the National Council for Gynecology and Fertility, issued a position paper referring to the vaccination of pregnant and lactating women, and to the concerns of a link between vaccinations and fertility. The title under which the Ministry of Health chose to keep the document was: “Vaccination for women and false fears of harming fertility.” This, even though the document itself explicitly states that “breastfeeding women were not included in the trials of the vaccine”. On what basis, then, did the Ministry of Health and the Council for Gynecology and Fertility decide to call the legitimate concerns of the public “false concerns”? The position paper does not explain, but states that, despite the lack of information, their “experts on this subject do not see a risk to the newborn from the breastfeeding mother’s vaccine and this is based on the accumulated experience of the use of vaccines against other viruses, the mechanism of action of the vaccine, the fact that there are no virus particles in the vaccine and the slim chance if any that the vaccine can reach the mother’s milk after its injection into the muscle”.
Another month later, in February 2021, the Ministry of Health published a press release entitled “Dr. Greenfeld: Answers to Questions, Rumors and Fake News About the Vaccine”, which included a video starring a family doctor from the Maccabi Health Fund, encouraging vaccination, including for pregnant and lactating women, and “apparently refutes” concerns. In relation to concerns about vaccination during breastfeeding, the doctor reassures in the video: “Just today a nursing patient of mine approached me and asked me, ‘Am I allowed to get vaccinated?’. I told her, ‘Yes, absolutely, and it is even recommended. In an examination carried out by doctors specializing in neonatal medicine, it was found that there is no fear of any side effect from the vaccine” (minute 3:38).
As noted above, the current analysis is a continuation of the analysis of the reports in the VAERS system of babies who themselves received the mRNA vaccines. Similar to the previous analysis, the data emerging from the current analysis constitute a red light that requires an in-depth examination of the potential effects these vaccines may have on babies when exposed to them via breast milk. Despite the large number of side effects reports found in our analysis, including significant side effects, the issue has hardly been discussed, so it is possible that many parents who experienced health problems in their breastfed babies were not aware of the possibility of a connection to the mother’s vaccine. Nevertheless, health authorities cannot claim that they did not know the data.
**By Ranit Feinberg
**Source

Warning: Please note that the chemical formula is HClO,not NaClO , and there is a problem with the translation !!! Ask the administrator to delete the following comments, thank you!!
The best way to solve the side effects of the new crown vaccine + The way to end 99% of the disease (Revised version)
Whether it is graphene oxide in the vaccine or the more horrible hydrogenated graphene, MMS2 generates hypochlorous acid, which can react to generate graphene and then be easily expelled from the body. Jim said that chlorine dioxide and hypochlorous acid can be naturally produced by the body’s immune system. MMS1 deals with spike protein, etc. To detoxify the vaccine, if it is serious, MMS 1+MMS 2 is the best. Self therapy requires self exploration (search for “the magic mineral solution of the third millennium”).
If MMS2 is taken too little, hypochlorous acid will be produced too little, with little effect; However, taking too much MMS2 powder will cause stomach pain and neutralize stomach acid too much, which is bad for the stomach. However, there is a perfect way to replace MMS2 powder, which is:
Non electrolytic pure hypochlorous acid solution (improved version of MMS2 is better than MMS1.)
Methods: The ratio of hypochlorous acid to water was 1:3, and at least 1L was drunk every day.
Note: 1.The chemical formula is HClO,not NaClO.
2. The water must be barreled or purified.
3. The temperature of water and hypochlorous acid must not exceed 40 ℃, and must be lower than 30 ℃, preferably 20 ℃. Because hypochlorous acid will decompose rapidly above 40 ℃.
The best way to solve the side effects of the new crown vaccine+the way to end 99% of the disease
Whether it is graphene oxide in the vaccine or the more horrible hydrogenated graphene, MMS2 generates hypochlorite, which can react to generate graphene and then be easily expelled from the body. Jim said that chlorine dioxide and hypochlorite can be naturally produced by the body’s immune system. MMS1 deals with spike protein, etc. It can detoxify cats. If it is serious, MMS1+MMS2 is the best. Self therapy requires self exploration (search for “the magic mineral solution of the third millennium”).
If MMS2 is taken too little, hypochlorite will be produced too little, with little effect; However, taking too much MMS2 powder will cause stomach pain and neutralize stomach acid too much, which is bad for the stomach. However, there is a perfect way to replace MMS2 powder, which is:
Non electrolytic pure hypochlorite solution (improved version of MMS2 is better than MMS1)
Methods: The ratio of hypochlorite to water was 1:3, and at least 1L was drunk every day.
Note: 1. The water must be barreled or purified.
2. The temperature of water and hypochlorite must not exceed 40 ℃, and must be lower than 30 ℃, preferably 20 ℃. Because hypochlorite will decompose rapidly above 40 ℃.
Please note that it is hypochlorous acid(HClO), not hypochlorite(NaClO). There is something wrong with Baidu translation.Please ask the administrator to delete this reply. Thank you
Warning: 1. The amount should not be too large. Excessive amount will cause great harm to the body. I feel very uncomfortable when I try 1.5-3L of solution for several days in a row. Most people should be sure to control it within an appropriate amount. If you are not sure, you should take MMS 2 according to Jim’s book.(If you have drunk a large amount of the solution and you feel unwell, please stop immediately and drink a lot of pure hot water.)
2.At present, some non electrolytic hypochlorous acid solutions also contain sodium hypochlorite, so before purchase, please be sure to ask the buyer whether it is pure hypochlorous acid, whether it contains sodium hypochlorite, and whether it can be mixed with water for drinking.
3.Water can be barreled water, mineral water or purified water, but do not add plasma water.